7 Key Principles: What Is an 8-Layer PCB Stackup and How to Design It Right
Discover what is an 8 layer pcb stackup, best layer configurations, materials, signal integrity stra
High-Reliability PCB/PCBA Manufacturing: Practical Guide to Design, Standards, Quality and Control
Table of Contents
ToggleSafety and clinical reliability are the core bottom lines of the medical device industry. As the core neural system of medical electronic devices, Printed Circuit Board Assemblies (PCBA) directly determine the operational stability of devices and even the lives of patients. Unlike consumer electronics and general industrial PCBA, medical device PCBA must adapt to the complex working environments of medical scenarios (e.g., high humidity, strong electromagnetic interference, long-term uninterrupted operation) and meet stringent global regulatory compliance requirements, making it a high-threshold niche track in the electronic manufacturing industry.
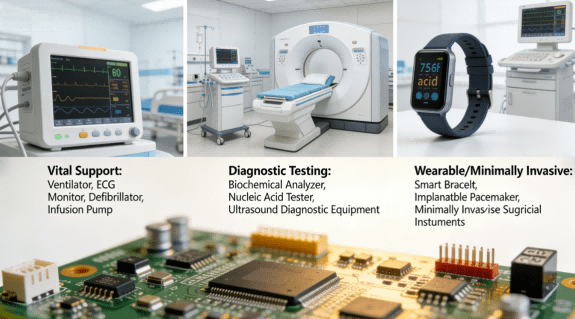
PCBA for medical devices is applied across all categories of medical electronics, mainly falling into three core types:
1.Life support equipment: Ventilators, electrocardiographic monitors, defibrillators, infusion pumps, etc., which require a Mission-critical Uptimeof over 99.99% for PCBA;
2.Diagnostic and testing equipment: Biochemical analyzers, nucleic acid detectors, ultrasonic diagnostic equipment, etc., which demand PCBA with high-precision signal transmission and anti-interference capabilities;
3.Wearable/minimally invasive devices: Smart bracelets, implantable pacemakers, minimally invasive surgical instruments, etc., which require miniaturized, highly integrated PCBA that meets biocompatibility requirements.
The global medical device contract manufacturing market size is projected to reach USD 131.36 billion in 2026, with a Compound Annual Growth Rate (CAGR) of 14.6%. Driven by population aging, the surging demand for chronic disease management and the transformation to high-precision and advanced manufacturing, China’s medical electronic market has become the core growth engine of the world. Meanwhile, the upgrading of medical devices toward miniaturization, wearability and AI intelligence has imposed more stringent technical requirements on PCBA manufacturing—0.4mm BGA pad pitch, ultra-miniature component mounting, and high-density interconnect (HDI) board processing have become industry standards, forcing manufacturing enterprises to enhance their core process capabilities.
Panoramic View Of Pcba Manufacturing Application Scenarios In Medical Devices
The manufacturing of medical device PCBA is not a mere process execution, but a full-process reliability control starting from the design stage. It must meet three-dimensional requirements of functional performance, compliance and environmental adaptability, with design and manufacturing standards far higher than those of general electronic products.
The design of medical PCBA adheres to the core principles of Design for Manufacturability (DFM), Design for Testability (DFT) and High Reliability, avoiding manufacturing defects and late operational risks in advance. The core design requirements include four aspects:
1.High-reliability hardware design: Prioritize PCB substrates with a high glass transition temperature (Tg ≥ 170℃) and low-loss dielectric materials (e.g., polyimide, high-performance FR-4, ceramic materials) to adapt to the thermal stability requirements of long-term uninterrupted operation of medical devices. For high-frequency diagnostic equipment, strictly control impedance matching (tolerance within ±5%) to ensure signal integrity and prevent electromagnetic interference from affecting detection accuracy.
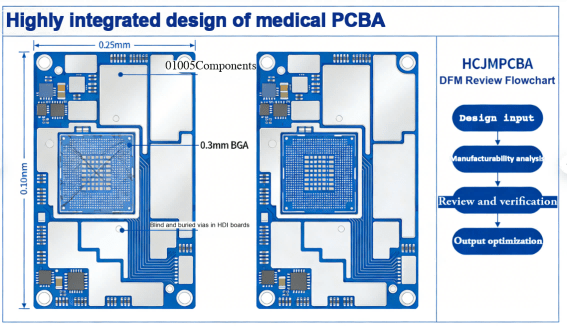
2.Ultra-miniaturization and high-integration design: Adapt to the size requirements of wearable and implantable medical devices, supporting the layout design of 01005 ultra-miniature componentsand 3mm pitch BGA. Meanwhile, rationally plan pad and trace spacing to avoid soldering short circuits, cold solder joints and other issues.
3.Design for Testability (DFT): Reserve sufficient In-Circuit Test (ICT) test points at the design stage with a 100% test point coverage rate, and adapt to the simulated working condition requirements of Functional Circuit Test (FCT). Ensure full-point detection in subsequent production to eliminate hidden faults.
4.Proactive Design for Manufacturability (DFM): Introduce manufacturing engineers for review at the initial design stage to put forward optimization suggestions for PCB layer count, component layout, soldering processes, etc., avoiding low production yield and poor late reliability caused by design defects. Huachuang Precision provides free proactive DFM reviewfor medical customers, resolving design defects before mass production.
Schematic Diagram Of High Integration Design For Medical Pcba
Material selection for medical PCBA directly determines the compliance and service life of products. It must meet three core requirements of industry standards, biocompatibility and environmental protection, and all materials must be traceable:
1.Medical-grade component selection: Prioritize automotive/medical-grade components and reject Commercial-grade components. Components must meet a wide temperature working range of -40℃~85℃. Meanwhile, establish a long-life cycle component management system to address End-of-Life (EOL) material issues. All materials are sourced from first-tier distributors such as Digi-Key and Mouser, with complete Certificate of Conformity (CoC)
2.Biocompatibility and environmental protection requirements: For medical PCBA in direct/indirect contact with patients, its substrate and coating materials must comply with the ISO 10993 biocompatibility standard, with no allergenic or toxic substances released. It also meets RoHS/REACH environmental protection requirements to adapt to global market access standards.
3.Application of protective materials: To withstand harsh hospital environments characterized by high humidity and aggressive disinfectants, conduct conformal coatingon PCBA with medical-grade conformal coating materials to improve the anti-pollution, anti-corrosion and moisture-proof capabilities of PCBA and avoid faults caused by environmental factors.
4.Lead-free soldering materials: Adopt medical-grade lead-free solder (Sn-Ag-Cu series)whose composition complies with the IPC J-STD-006 standard, ensuring the mechanical strength and oxidation resistance of solder joints and avoiding equipment faults caused by solder joint aging.

Hcjmpcba Medical Grade Material Warehouse Live Shot
The manufacturing process of medical PCBA takes IPC Class 3 (the highest reliability grade for electronic manufacturing) as the implementation standard. For the processing of ultra-miniature components and high-density boards, it achieves refined and standardized process control. The core process requirements include:
1.High-precision SMT mounting process: Equipped with high-precision pick-and-place machines to realize accurate mounting of 01005 packaged components and 0.3mm pitch BGA with a mounting accuracy of ±0.02mm, avoiding component offset, tombstoning and other defects. Conduct plasma cleaning on PCB before mounting to improve pad wettability and reduce the risk of cold solder joints.
2.Refined reflow soldering process: Adopt hot air reflow soldering and customize exclusive temperature profiles for different components and PCB thicknesses to achieve zoned temperature control (temperature difference ±2℃), avoiding component damage and PCB warpage caused by excessively high temperature, or cold solder joints and solder joint voids caused by excessively low temperature. For packaged components such as BGA and QFN, inspect the internal quality of solder joints through X-Ray after soldering, with the void rate controlled within 5%(far lower than the 20% standard of IPC Class 3).
3.Multi-layer HDI board processing process: Proficiency in the assembly process of 8-32 layer HDI high-density interconnect boards, with precise control of blind/buried via drilling and electroplating accuracy to meet the high-speed signal transmission requirements of medical imaging and high-end diagnostic equipment, and the board thickness tolerance controlled within ±0.05mm.
4.Manual insertion and soldering process: For the insertion of special-shaped components and high-power components, IPC-certified technicianswith constant temperature soldering irons. 100% visual inspection is conducted after soldering to ensure that the verticality of inserted components and the fullness of solder joints comply with the IPC Class 3 standard.

Hcjmpcba High Precision Smt Mounting Production Line Live Shot
2026 is a watershed for global medical regulation. The US FDA Quality Management System Regulation (QMSR) is fully integrated with the ISO 13485:2016 standard, and PCBA manufacturing enterprises without ISO 13485 certification will be completely excluded from the global high-end medical supply chain. The compliance of medical device PCBA is not the satisfaction of a single standard, but the dual compliance of global regulatory standards and electronic manufacturing standards. The core standard system is as follows:
ISO 13485 is a globally recognized quality management system standard for the medical device industry. Different from the general ISO 9001, it focuses on the safety and effectiveness of medical devices, with more stringent and targeted requirements for PCBA manufacturing, and the risk management principle runs through the entire process.
In medical PCBA manufacturing, the core implementation requirements of ISO 13485 include:
1.Full-process risk management coverage: Introduce Failure Mode and Effects Analysis (FMEA)from design review, material procurement, production and manufacturing to finished product delivery, conduct risk assessment for each process and each component, identify potential failure points and formulate preventive measures in advance;
2.Full-life cycle traceability: Realize board-level two-way traceabilityfrom component batch, production station, equipment parameters to finished product serial number. In the event of a product recall, the affected batches can be accurately located within a few hours;
3.Process validation and revalidation: Conduct strict Installation Qualification (IQ), Operational Qualification (OQ)and Performance Qualification (PQ) for processes that cannot be fully verified by non-destructive testing (e.g., reflow soldering, conformal coating). Revalidation is required when processes, equipment or materials change to ensure process stability;
4.Complete document record system: Standardized documents must be formed for the operating specifications, test data and personnel information of all processes, with a retention period not less than the service life of medical devicesto meet the audit requirements of regulatory authorities.
Huachuang Precision has obtained dual certification of ISO 13485:2016 and ISO 9001, and integrated the requirements of ISO 13485 into the entire production process, becoming a benchmark for compliant execution in medical PCBA manufacturing.
IPC standards are universal standards for the global electronic manufacturing industry, among which IPC Class 3 is the highest reliability grade and the minimum implementation standard for medical device PCBA. Its core requirements focus on the long-term fault-free operation capability of products, and the core implementation clauses include:
1.100% full inspection requirement: All PCBA must undergo visual inspection, 3D Automated Optical Inspection (AOI), 3D Solder Paste Inspection (SPI) and X-Ray inspection, with no solder abnormalities, component offset, solder joint voids and other defects;
2.Stringent solder joint quality requirements: Solder joints must be full, free of cold solder joints, microcracks and bridges. The BGA solder joint void rate is ≤20%, and Huachuang Precision has an internal control index of ≤5%, far exceeding the industry standard;
3.Material cleanliness requirement: The ionic contamination of the PCBA surface is ≤1.5μgNaCl/cm²to avoid circuit board corrosion and short circuits caused by contaminants;
4.Mechanical strength requirement: The mechanical strength of components after soldering must pass tensile and vibration tests to avoid component falling off caused by vibration during transportation and equipment operation.
In addition to IPC Class 3, medical PCBA manufacturing must also comply with special standards such as IPC-A-610 (Acceptability of Electronic Assemblies), IPC-A-6012 (Performance Specification for Rigid Printed Boards) and IPC J-STD-001 (Requirements for Soldering Electrical and Electronic Assemblies), forming a complete electronic manufacturing standard system.
Medical PCBA must adapt to the regulatory requirements of target markets, and the core regional standards include:
1.US market: FDA CFR 21 Part 820 (Quality System Regulation for Medical Devices), which requires PCBA manufacturing enterprises to establish a sound quality system, and products must be registered with the FDA, with relevant test data meeting FDA audit requirements;
2.EU market: Medical Device Regulation (MDR), which requires PCBA to meet the dual requirements of “clinical evidence + compliance system”, and obtain CE certification to meet the EU’s product traceability and adverse event reporting requirements;
3.Global universal: IEC 60601 series standards (Safety of Medical Electrical Equipment), which require PCBA to have safety performance such as electric shock protection, electromagnetic interference protection and overheating protection, adapting to the electrical safety requirements of medical devices.

Hcjmpcba Iso 13485
Quality control of medical device PCBA is not “finished product inspection”, but a closed-loop management system from the supply chain end to the delivery end. Its core goals are to achieve zero-defect manufacturing and full-process traceability, while meeting the audit requirements of regulatory authorities. Combining the requirements of ISO 13485 and the pain points of the medical industry, Huachuang Precision has established an exclusive quality management and traceability system for medical PCBA, which includes five core modules:
Materials are the foundation of PCBA quality. Material control in the medical supply chain must achieve three goals: authentic products, traceability and stable supply:
1.Qualified supplier access: Establish a medical-grade supplier review system, cooperate only with original manufacturers and first-tier distributors, conduct multi-dimensional audits of suppliers on ISO 13485, quality system, delivery capacity, etc., and sign exclusive quality agreements;
2.Incoming Quality Control (IQC): After all materials arrive at the factory, conduct a full-item inspection through component testing equipment and visual inspection equipment to verify material model, batch, appearance and electrical performance, eliminating counterfeit, refurbished and unqualified materials from entering the production process;
3.Material batch management: All materials are affixed with a unique batch code, and a material batch ledger is established to realize batch association of materials from warehousing, collection, production to finished products, ensuring material traceability;
4.Long-life cycle material management: In view of the long R&D and mass production cycles of medical devices, establish an End-of-Life (EOL) material early warning systemto provide customers with material replacement solutions in advance and avoid project stagnation caused by material discontinuation.
The production process is the core of quality control. A three-level inspection mode of first article inspection + in-process inspection + full inspection is adopted to ensure the quality stability of each process:
1.First article confirmation: Before the production of each batch, make a first article PCBA and conduct a full-item confirmation through ICT, visual inspection and X-Ray inspection. Mass production can only be carried out after the first article is qualified;
2.In-Process Quality Control (IPQC): Establish a 3-time quality inspection system. Engineers conduct in-process inspections of production processes every 30 minutes, record equipment parameters, process execution and product quality, and timely detect and correct process deviations;
3.Process special inspection: Conduct 3D SPI and 3D AOI inspection after SMT mounting, X-Ray inspection after reflow soldering, 100% visual inspection after insertion soldering, and coating thickness inspection after conformal coating. Each process can only enter the next one after passing the inspection;
4.Final Quality Control (FQC): After the finished product is completed, conduct ICT, FCT and full visual inspection. All test data are uploaded to the system in real time to form a finished product inspection file. Non-conforming products are directly isolated to prevent defective products from flowing out.
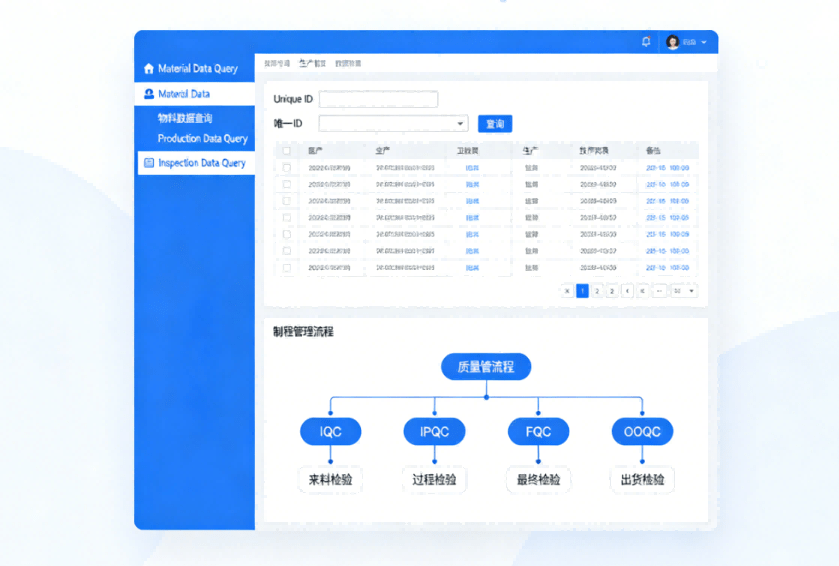
In accordance with the traceability requirements of ISO 13485, assign a unique identity ID to each medical PCBA and establish a two-way traceability system from “materials” to “finished products”. The traceability information includes:
1.Material information: Component model, batch, supplier, incoming inspection report;
2.Production information: Production station, equipment model, process parameters, operator, production time;
3.Test information: Test data of each process, test equipment, tester, non-conforming item processing record;
4.Delivery information: Finished product serial number, customer delivered to, delivery time, logistics information.
Through the traceability system, two-way queries of “checking material batches by finished product ID” and “checking finished product scope by material batch” can be realized, meeting the customer’s quality traceability and regulatory authority audit requirements.
Establish a sound quality document system. All documents are compiled, approved and updated in accordance with the requirements of ISO 13485. The core documents include: quality manual, procedure documents, work instructions, test standards, batch records, test reports, etc. All documents are managed electronically with a retention period not less than the service life of medical devices, and support online query and printing to meet the document retrieval requirements of regulatory audits.
Establish a strict non-conforming product control process. For non-conforming products found in the production process, implement a closed-loop management of isolation – identification – analysis – processing – verification:
1.Physically isolate non-conforming products and affix identification labels, specifying the non-conforming reason, batch and quantity;
2.Organize engineers and process engineers to conduct root cause analysis to identify the root cause of non-conformity;
3.Formulate corrective and preventive measures, such as optimizing process parameters, replacing materials, and training employees;
4.Verify the implementation effect of the measures to ensure that similar problems do not recur;
5.Processing records of all non-conforming products are formed into documents and incorporated into the quality system for continuous improvement.

Hcjmpcba Medical Pcba Testing Laboratory Live Shot
Testing and validation of medical PCBA is the last line of defense for quality control and an important basis for products to obtain market access. It must realize full-dimensional validation of electrical performance, functional performance, environmental adaptability and safety performance. All test data must form standardized reports as the technical basis for customer product registration. Huachuang Precision has established an exclusive testing laboratory for medical PCBA, equipped with industry-leading testing equipment to achieve full coverage of testing. The core testing and validation processes are as follows:
Core testing equipment: 3D AOI, 3D SPI, X-Ray inspection equipment, ICT In-Circuit Tester
1.Solder Paste Inspection (SPI): Before SMT mounting, inspect the printing thickness, area and position of solder paste to ensure that solder paste printing complies with process requirements and avoid cold solder joints and short circuits caused by solder paste problems;
2.Automated Optical Inspection (AOI): After mounting and soldering, detect appearance defects such as component offset, tombstoning, solder bridging and insufficient solder through 3D AOI with a detection accuracy of 0.01mm;
3.X-Ray inspection: For hidden solder joints such as BGA and QFN, detect internal defects such as voids, cracks and cold solder joints in solder joints through X-Ray to ensure the internal quality of solder joints;
4.In-Circuit Test (ICT): Contact the test points of PCBA through a needle bed to detect electrical parameters such as on-off, resistance, capacitance and inductance of the circuit, ensure no circuit connection faults with a 100% detection coverage rate.
Core testing equipment: FCT functional test bench, signal analyzer, oscilloscope
1.Functional Circuit Test (FCT): Build an exclusive FCT test bench according to the working conditions of medical equipment provided by customers, simulate the actual operating environment of the equipment, and test various functional parameters of PCBA (e.g., signal transmission, data processing, instruction execution) to ensure that the performance of PCBA meets the standards under actual working conditions;
2.High and low temperature functional test: Conduct high and low temperature functional tests in a temperature chamber at -40℃~85℃ to verify the functional stability of PCBA in a wide temperature environment and avoid performance abnormalities caused by temperature changes;
3.Signal integrity test: For PCBA of high-frequency diagnostic equipment, detect the amplitude, frequency and delay of signal transmission through signal analyzers and oscilloscopes to ensure no signal distortion or interference.
Core testing equipment: Temperature and humidity cycle test chamber, vibration test bench, salt spray test chamber
1.Temperature and humidity cycle test: Conduct temperature and humidity cycle tests at -40℃85℃ and 20%95% humidity in accordance with the IEC 60068 standard with more than 1000 cyclesto verify the long-term stability of PCBA in an environment with temperature and humidity changes;
2.Vibration test: Simulate the vibration environment of medical equipment during transportation and use, conduct vibration tests at a frequency of 5~2000Hz to verify the mechanical strength of PCBA solder joints and the fixing stability of components;
3.Salt spray test: In response to the high-humidity and corrosive environment of hospitals, conduct a neutral salt spray test (5%NaCl, temperature 35℃)for more than 48 hours to verify the corrosion resistance of PCBA.
Core testing equipment: Withstand voltage tester, insulation resistance tester, Electromagnetic Compatibility (EMC) test system
1.Withstand voltage test: Detect the insulation performance of PCBA to avoid electric shock risks caused by poor insulation, and the withstand voltage value complies with the IEC 60601 standard;
2.Insulation resistance test: Test the insulation resistance between each circuit of PCBA and between the circuit and the shell to ensure that the insulation resistance ≥100MΩ, meeting the electrical safety requirements of medical devices;
3.EMC electromagnetic compatibility test: Test the electromagnetic radiation and anti-interference capabilities of PCBA, complying with the YY 0505 standard (Electromagnetic Compatibility Requirements for Medical Electrical Equipment), to prevent electromagnetic interference generated by PCBA from affecting other equipment or causing performance abnormalities due to interference from other equipment.
After all tests are completed, provide customers with standardized test reports including test items, test equipment, test parameters, test results, judgment conclusions and other information. The reports are associated with the unique ID of PCBA to realize the traceability of test data. The test reports can be directly used as the technical basis for customer medical device product registration and clinical certification, helping customers accelerate the speed of market access.
Screenshot Of Hcjmpcba Medical Pcba Traceability Management System Interface
The manufacturing of medical device PCBA is a dynamic process. Minor changes in processes, equipment and materials may bring quality risks. Therefore, it is necessary to establish a management system of traceability visualization + proactive risk control to realize real-time monitoring and risk early warning of the entire production process. The core includes three key measures:
Build a digital production management system, connect all production and testing equipment to the system to realize real-time data collection, real-time status monitoring and real-time abnormal alarm of the production process:
1.The system displays the production progress, equipment operation parameters, process execution and test qualification rate of each production line in real time;
2.When equipment parameters deviate from process requirements, test qualification rate is lower than the threshold, or non-conforming products appear, the system immediately issues an audible and visual alarm, and engineers intervene in the first time;
3.Production data is uploaded to the cloud in real time, and customers can view the production progress and test data through an exclusive port to realize transparent supply chain management.
Establish a strict ECN process for design, material and process changes to ensure that all changes are implemented after review, verification and approval, avoiding quality risks caused by improper changes:
1.Change application: The customer or internal engineer submits an ECN application, explaining the change reason, content and scope of influence;
2.Change review: Organize the design, process, quality, production and other departments to conduct a review to evaluate the impact of the change on product quality, compliance and production processes;
3.Change verification: Make samples for the change content and conduct a full-item test verification. The change can only be approved after the verification is qualified;
4.Change execution: Issue an ECN notice to all relevant departments, update documents such as work instructions and test standards, and conduct change training for employees to ensure the implementation of the change;
5.Change record: All ECN application, review, verification and execution records are formed into documents and incorporated into the quality system to realize the traceability of changes.
1.Failure Mode and Effects Analysis (FMEA): At the product R&D and production planning stage, organize a cross-departmental team to conduct FMEA analysis, identify potential failure points in product design and production processes, evaluate the severity, occurrence probability and detectability of failures, formulate risk reduction measures, and resolve risks in the proactive stage;
2.Risk early warning system: Establish a risk early warning system for materials, equipment and processes to provide early warning of potential risks such as material discontinuation, equipment failure and process parameter deviation, and formulate emergency plans to ensure the continuity and stability of production.
As a professional PCBA manufacturing enterprise rooted in Guangzhou and radiating the world, Huachuang Precision focuses on the medical electronics field, with the ISO 13485 system as the core, and integrates three capabilities of high-precision processes, digital management and customized services. It has become a core supply chain partner for medical OEM enterprises and one of the few enterprises in China that can realize full-process compliant manufacturing of medical PCBA. The core advantages include four dimensions:
1.Obtained dual certification of ISO 13485:2016 and ISO 9001 quality management systems, with the entire production process complying with the requirements of FDA QMSR and EU MDR;
2.Strictly implement the IPC Class 3 electronic manufacturing standard, with internal control indicators far exceeding industry standards (e.g., BGA solder joint void rate ≤5%);
3.Establish a sound traceability system and quality document system to meet the audit requirements of regulatory authorities and the product registration requirements of customers.
1.Ultra-precision mounting: Support the mounting of 01005 packaged components and 0.3mm pitch BGA with a mounting accuracy of ±0.02mm, adapting to the miniaturization and high-integration requirements of medical devices;
2.High-density board processing: Proficiency in the assembly process of 8-32 layer HDI boards, with precise control of blind/buried via processing and high-speed signal transmission to meet the requirements of high-end diagnostic equipment;
3.Full coverage of testing: Equipped with a complete set of testing equipment such as 3D AOI, 3D SPI, X-Ray, ICT/FCT to realize full-process testing from solder paste printing to finished product delivery;
4.Intelligent manufacturing: Build a digital production management system and traceability system to realize visualization, digitalization and traceability of the production process.
1.Medical Green Channel Production Line: Set up an exclusive production line for medical PCBA with no queuing for small-batch production. Prototype samples can be delivered in as fast as 24 hours, solving the pain points of fast R&D iteration and slow sample delivery for medical enterprises and helping customers accelerate clinical certification progress;
2.Free DFM Review: Provide professional DFM engineer review for customers before mass production, correct design defects and improve the first article qualification rate of products to over 99.5%;
3.Long-life cycle material management: Establish an EOL material early warning system to provide customers with material replacement solutions and solve the core pain point of material discontinuation in the medical industry;
4.24H Rapid Response: Establish an exclusive service team for medical customers to respond to customer needs 24/7 and timely solve problems in R&D, production and delivery.
1.Relying on the electronic manufacturing industrial belt in the Pearl River Delta, establish long-term cooperation with first-tier distributors such as Digi-Key and Mouser to realize the genuine product guarantee and stable supply of medical-grade components;
2.Establish a 10,000-class material warehouse to realize spot inventory of commonly used medical components, shorten the procurement cycle and improve delivery efficiency;
3.Possess strong supply chain integration capabilities, and can provide customers with one-stop services of PCB + PCBA + component procurementto reduce the customer’s supply chain management costs.

Hcjmpcba Medical Pcba Exclusive Green Channel Production Line Live Shot
With the in-depth transformation of global medical electronics toward miniaturization, wearability, AI intelligence and minimal invasion, as well as the further integration of ISO 13485 with global regulatory standards, the medical device PCBA industry will witness three core changes from 2026 to 2030. Huachuang Precision has made advance layout to build core technical competitiveness and adapt to the future development of the industry:
With the popularization of wearable medical devices (e.g., smart ECG bracelets, blood glucose monitors) and minimally invasive surgical instruments, the demand for Flexible Printed Circuits (FPC) and rigid-flex PCBs will experience explosive growth, requiring PCBA manufacturing enterprises to have the mounting, soldering and protective process capabilities for flexible boards.
Huachuang Precision’s Layout: Has introduced exclusive production equipment and processes for flexible boards to realize high-precision mounting and conformal coating of FPC/rigid-flex PCBs, adapting to the size and performance requirements of wearable and implantable medical devices.
The application of AI technology in medical diagnosis and life support is deepening, and medical devices are upgrading to edge AI. This requires PCBA to integrate higher-performance computing units and sensors, and at the same time puts forward higher requirements for PCBA’s heat dissipation management, signal integrity and anti-interference capabilities.
Huachuang Precision’s Layout: Set up a R&D team for high-end high-speed PCBA, conduct in-depth research on heat dissipation design and high-frequency signal impedance control technology, realize accurate assembly of high-performance computing units and customized heat dissipation solutions to meet the requirements of edge AI medical devices.
The medical industry has increasingly high requirements for “zero defect” of PCBA. Traditional manual inspection can no longer meet the inspection needs of ultra-miniaturized and highly integrated PCBA. AI-driven visual inspection and fully automated production will become the industry mainstream.
Huachuang Precision’s Layout: Continuously invest in intelligent equipment, introduce AI visual inspection systems, fully automated mounting lines and unmanned inspection lines to realize full automation of production and inspection. At the same time, combine big data analysis to realize early warning of quality risks and build a genuine “zero-defect manufacturing” system.
The manufacturing of medical device PCBA is a comprehensive embodiment of technology, standards, compliance and services. Its core is not mere process execution, but full-process high-reliability control from the design stage to the delivery stage, as well as an in-depth understanding and implementation of global medical regulatory standards. Against the backdrop of the full integration of FDA QMSR and ISO 13485 in 2026, compliance has become an entry ticket for medical PCBA manufacturing enterprises, while high-precision processes, digital management and customized services have become the core competitiveness of enterprises.
Guangzhou Huachuang Precision Technology Co., Ltd. (HCJMPCBA), as a professional service provider of medical device PCBA manufacturing, takes the ISO 13485 system as its gene and integrates high-reliability manufacturing into every process. We provide medical OEM enterprises with one-stop solutions from PCB design, material procurement, PCBA manufacturing to testing and validation, solving the core pain points of slow R&D iteration, high compliance difficulty and unstable supply chain for customers.
In the medical field, quality is not an option, but a bottom line. Choosing Huachuang Precision means choosing a medical PCBA supply chain partner who is well-versed in global regulatory standards, has core process capabilities and can cooperate for a long time. We will help medical enterprises enhance their core competitiveness with high-reliability, high-compliance and high-customized products and services, and jointly promote the development of the medical electronics industry.
Free DFM Review: Contact Huachuang Precision engineers for professional DFM review services for your medical PCBA design to avoid design defects in advance.
Rapid Sample Delivery: Medical prototype samples can be delivered in as fast as 24 hours to facilitate R&D iteration and clinical certification.
Obtain Compliance Data Package: Receive exclusive materials such as ISO 13485 Compliance Execution Manual, Medical PCBA Manufacturing Standards and Test Report Samples.
Official Website: hcjm-pcba.com
Company Address: Guangzhou City, Guangdong Province, China
Tel(WhatsApp):86-13660883282
Discover what is an 8 layer pcb stackup, best layer configurations, materials, signal integrity stra
For PCB/PCBA OEM engineers, procurement decision-makers, and cross-industry buyers, every component
Discover what a 10k resistor is, how to read its color bands, and why this fundamental component is